Hard sphere model: Difference between revisions
Carl McBride (talk | contribs) m (Added code link to SMAC-wiki.) |
(Fixed broken links in ref. 5) |
||
| (77 intermediate revisions by 14 users not shown) | |||
| Line 1: | Line 1: | ||
[[Image:sphere_green.png|thumb|right]] | [[Image:sphere_green.png|thumb|right]] | ||
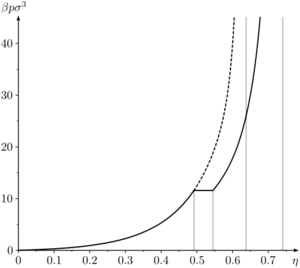
[[Image:Hard-sphere phase diagram pressure vs packing fraction.png|thumb|right|Phase diagram (pressure vs packing fraction) of hard sphere system (Solid line - stable branch, dashed line - metastable branch)]] | |||
The hard sphere | The '''hard sphere model''' (sometimes known as the ''rigid sphere model'') is defined as | ||
: <math> | : <math> | ||
\ | \Phi_{12}\left( r \right) = \left\{ \begin{array}{lll} | ||
\infty & ; & r < \sigma \\ | \infty & ; & r < \sigma \\ | ||
0 & ; & r \ge \sigma \end{array} \right. | 0 & ; & r \ge \sigma \end{array} \right. | ||
</math> | </math> | ||
where <math> \ | where <math> \Phi_{12}\left(r \right) </math> is the [[intermolecular pair potential]] between two spheres at a distance <math>r := |\mathbf{r}_1 - \mathbf{r}_2|</math>, and <math> \sigma </math> is the diameter of the sphere. | ||
The hard sphere model can be considered to be a special case of the [[hard ellipsoid model]], where each of the semi-axes has the same length, <math>a=b=c</math>. | |||
==First simulations of hard spheres (1954-1957)== | |||
The hard sphere model, along with its two-dimensional manifestation [[hard disks]], was one of the first ever systems studied using [[computer simulation techniques]] with a view | |||
to understanding the thermodynamics of the liquid and solid phases and their corresponding [[Phase transitions | phase transition]] | |||
<ref>[http://dx.doi.org/10.1063/1.1740207 Marshall N. Rosenbluth and Arianna W. Rosenbluth "Further Results on Monte Carlo Equations of State", Journal of Chemical Physics '''22''' pp. 881-884 (1954)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.1743956 W. W. Wood and J. D. Jacobson "Preliminary Results from a Recalculation of the Monte Carlo Equation of State of Hard Spheres", Journal of Chemical Physics '''27''' pp. 1207-1208 (1957)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.1743957 B. J. Alder and T. E. Wainwright "Phase Transition for a Hard Sphere System", Journal of Chemical Physics '''27''' pp. 1208-1209 (1957)]</ref>, much of this work undertaken at the Los Alamos Scientific Laboratory on the world's first electronic digital computer ENIAC <ref>[http://ftp.arl.army.mil/~mike/comphist/eniac-story.html The ENIAC Story]</ref>. | |||
==Liquid phase radial distribution function== | |||
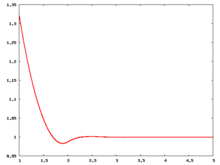
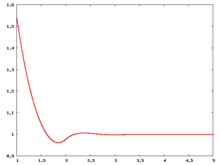
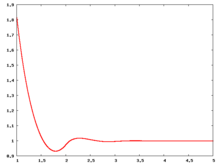
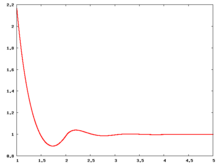
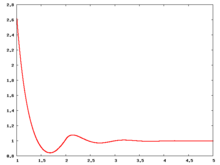
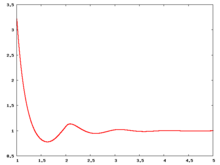
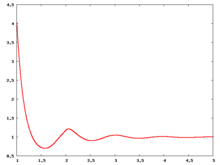
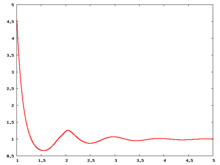
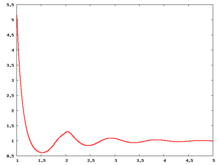
The following are a series of plots of the hard sphere [[radial distribution function]] <ref>The [[total correlation function]] data was produced using the [https://old.vscht.cz/fch/software/hsmd/hspline-8-2004.zip computer code] written by [https://web.vscht.cz/~kolafaj/ Jiří Kolafa]</ref> shown for different values of the number density <math>\rho</math>. The horizontal axis is in units of <math>\sigma</math> where <math>\sigma</math> is set to be 1. Click on image of interest to see a larger view. | |||
:{| border="1" | |||
|- | |||
|<math>\rho=0.2</math> [[Image:HS_0.2_rdf.png|center|220px]] ||<math>\rho=0.3</math> [[Image:HS_0.3_rdf.png|center|220px]] || <math>\rho=0.4</math> [[Image:HS_0.4_rdf.png|center|220px]] | |||
|- | |||
|<math>\rho=0.5</math> [[Image:HS_0.5_rdf.png|center|220px]] ||<math>\rho=0.6</math> [[Image:HS_0.6_rdf.png|center|220px]] || <math>\rho=0.7</math> [[Image:HS_0.7_rdf.png|center|220px]] | |||
|- | |||
|<math>\rho=0.8</math> [[Image:HS_0.8_rdf.png|center|220px]] ||<math>\rho=0.85</math> [[Image:HS_0.85_rdf.png|center|220px]] || <math>\rho=0.9</math> [[Image:HS_0.9_rdf.png|center|220px]] | |||
|} | |||
The value of the radial distribution at contact, <math>{\mathrm g}(\sigma^+)</math>, can be used to calculate the [[pressure]] via the [[equations of state |equation of state]] (Eq. 1 in <ref name="Tao1"> [http://dx.doi.org/10.1103/PhysRevA.46.8007 Fu-Ming Tao, Yuhua Song, and E. A. Mason "Derivative of the hard-sphere radial distribution function at contact", Physical Review A '''46''' pp. 8007-8008 (1992)]</ref>) | |||
:<math>\frac{p}{\rho k_BT}= 1 + B_2 \rho {\mathrm g}(\sigma^+)</math> | |||
where the [[second virial coefficient]], <math>B_2</math>, is given by | |||
:<math>B_2 = \frac{2\pi}{3}\sigma^3</math>. | |||
Carnahan and Starling <ref>[http://dx.doi.org/10.1063/1.1672048 N. F.Carnahan and K. E.Starling,"Equation of State for Nonattracting Rigid Spheres" Journal of Chemical Physics '''51''' pp. 635-636 (1969)]</ref> provided the following expression for <math>{\mathrm g}(\sigma^+)</math> (Eq. 3 in <ref name="Tao1" ></ref>) | |||
:<math>{\mathrm g}(\sigma^+)= \frac{1-\eta/2}{(1-\eta)^3}</math> | |||
where <math>\eta</math> is the [[packing fraction]]. | |||
Over the years many groups have studied the radial distribution function of the hard sphere model: | |||
<ref>[http://dx.doi.org/10.1063/1.1747854 John G. Kirkwood, Eugene K. Maun, and Berni J. Alder "Radial Distribution Functions and the Equation of State of a Fluid Composed of Rigid Spherical Molecules", Journal of Chemical Physics '''18''' pp. 1040- (1950)]</ref> | |||
<ref>[http://dx.doi.org/10.1103/PhysRev.85.777 B. R. A. Nijboer and L. Van Hove "Radial Distribution Function of a Gas of Hard Spheres and the Superposition Approximation", Physical Review '''85''' pp. 777 - 783 (1952)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.1742004 B. J. Alder, S. P. Frankel, and V. A. Lewinson "Radial Distribution Function Calculated by the Monte-Carlo Method for a Hard Sphere Fluid", Journal of Chemical Physics '''23''' pp. 417- (1955)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.1727245 Francis H. Ree, R. Norris Keeler, and Shaun L. McCarthy "Radial Distribution Function of Hard Spheres", Journal of Chemical Physics '''44''' pp. 3407- (1966)]</ref> | |||
<ref>[http://dx.doi.org/10.1080/00268977000101421 W. R. Smith and D. Henderson "Analytical representation of the Percus-Yevick hard-sphere radial distribution function", Molecular Physics '''19''' pp. 411-415 (1970)]</ref> | |||
<ref>[http://dx.doi.org/10.1080/00268977100101331 J. A. Barker and D. Henderson "Monte Carlo values for the radial distribution function of a system of fluid hard spheres", Molecular Physics '''21''' pp. 187-191 (1971)]</ref> | |||
<ref>[http://dx.doi.org/10.1080/00268977700102241 J. M. Kincaid and J. J. Weis "Radial distribution function of a hard-sphere solid", Molecular Physics '''34''' pp. 931-938 (1977)]</ref> | |||
<ref>[http://dx.doi.org/10.1103/PhysRevA.43.5418 S. Bravo Yuste and A. Santos "Radial distribution function for hard spheres", Physical Review A '''43''' pp. 5418-5423 (1991)]</ref> | |||
<ref>[http://dx.doi.org/10.1080/00268979400100491 Jaeeon Chang and Stanley I. Sandler "A real function representation for the structure of the hard-sphere fluid", Molecular Physics '''81''' pp. 735-744 (1994)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.1979488 Andrij Trokhymchuk, Ivo Nezbeda and Jan Jirsák "Hard-sphere radial distribution function again", Journal of Chemical Physics '''123''' 024501 (2005)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.2201699 M. López de Haro, A. Santos and S. B. Yuste "On the radial distribution function of a hard-sphere fluid", Journal of Chemical Physics '''124''' 236102 (2006)]</ref> | |||
==Liquid-solid transition== | |||
The hard sphere system undergoes a [[Solid-liquid phase transitions |liquid-solid]] [[First-order transitions |first order transition]] <ref name="HooverRee">[http://dx.doi.org/10.1063/1.1670641 William G. Hoover and Francis H. Ree "Melting Transition and Communal Entropy for Hard Spheres", Journal of Chemical Physics '''49''' pp. 3609-3617 (1968)]</ref> | |||
<ref>[http://dx.doi.org/10.1063/1.4870524 Miguel Robles, Mariano López de Haro and Andrés Santos "Note: Equation of state and the freezing point in the hard-sphere model", Journal of Chemical Physics '''140''' 136101 (2014)]</ref>, sometimes referred to as the Kirkwood-Alder transition <ref name="GastRussel">[http://dx.doi.org/10.1063/1.882495 Alice P. Gast and William B. Russel "Simple Ordering in Complex Fluids", Physics Today '''51''' (12) pp. 24-30 (1998)]</ref>. | |||
The liquid-solid coexistence densities (<math>\rho^* = \rho \sigma^3=6\eta/\pi</math>) has been calculated to be | |||
:{| border="1" | |||
|- | |||
| <math>\rho^*_{\mathrm {solid}}</math> || <math>\rho^*_{\mathrm {liquid}}</math> || Reference | |||
|- | |||
| 1.041(4)|| 0.943(4) || <ref name="HooverRee"></ref> | |||
|- | |||
| 1.0376|| 0.9391 || <ref name="FrenkelSmitBook">Daan Frenkel and Berend Smit "Understanding Molecular Simulation: From Algorithms to Applications", Second Edition (2002) (ISBN 0-12-267351-4) p. 261.</ref> | |||
|- | |||
| 1.0367(10) || 0.9387(10) || <ref name="Fortini">[http://dx.doi.org/10.1088/0953-8984/18/28/L02 Andrea Fortini and Marjolein Dijkstra "Phase behaviour of hard spheres confined between parallel hard plates: manipulation of colloidal crystal structures by confinement", Journal of Physics: Condensed Matter '''18''' pp. L371-L378 (2006)]</ref> | |||
|- | |||
| 1.0372 || 0.9387 || <ref name="VegaNoya"> [http://dx.doi.org/10.1063/1.2790426 Carlos Vega and Eva G. Noya "Revisiting the Frenkel-Ladd method to compute the free energy of solids: The Einstein molecule approach", Journal of Chemical Physics '''127''' 154113 (2007)]</ref> | |||
|- | |||
| 1.0369(33) || 0.9375(14) || <ref name="Noya"> [http://dx.doi.org/10.1063/1.2901172 Eva G. Noya, Carlos Vega, and Enrique de Miguel "Determination of the melting point of hard spheres from direct coexistence simulation methods", Journal of Chemical Physics '''128''' 154507 (2008)]</ref> | |||
|- | |||
| 1.037 || 0.938 || <ref>[http://dx.doi.org/10.1063/1.476396 Ruslan L. Davidchack and Brian B. Laird "Simulation of the hard-sphere crystal–melt interface", Journal of Chemical Physics '''108''' pp. 9452-9462 (1998)]</ref> | |||
|- | |||
| 1.033(3) || 0.935(2) || <ref name="Miguel"> [http://dx.doi.org/10.1063/1.3023062 Enrique de Miguel "Estimating errors in free energy calculations from thermodynamic integration using fitted data", Journal of Chemical Physics '''129''' 214112 (2008)]</ref> | |||
|- | |||
| 1.03715(9) || 0.93890(7) || <ref name="MoirEtAl2021"> [https://doi.org/10.1063/5.0058892 Craig Moir, Leo Lue, and Marcus N. Bannerman "Tethered-particle model: The calculation of free energies for hard-sphere systems", Journal of Chemical Physics '''155''' 064504 (2021)]</ref> | |||
|} | |||
The coexistence [[pressure]] has been calculated to be | |||
:{| border="1" | |||
|- | |||
| <math>p (k_BT/\sigma^3) </math> || Reference | |||
|- | |||
| 11.5727(10)|| <ref name="FernandezUCM">[http://dx.doi.org/10.1103/PhysRevLett.108.165701 L. A. Fernández, V. Martín-Mayor, B. Seoane, and P. Verrocchio "Equilibrium Fluid-Solid Coexistence of Hard Spheres", Physical Review Letters '''108''' 165701 (2012)]</ref> | |||
|- | |||
| 11.57(10) || <ref name="Fortini"></ref> | |||
|- | |||
| 11.567|| <ref name="FrenkelSmitBook"></ref> | |||
|- | |||
| 11.55(11) || <ref>[http://dx.doi.org/10.1088/0953-8984/9/41/006 Robin J. Speedy "Pressure of the metastable hard-sphere fluid", Journal of Physics: Condensed Matter '''9''' pp. 8591-8599 (1997)]</ref> | |||
|- | |||
| 11.54(4) || <ref name="Noya"></ref> | |||
|- | |||
| 11.50(9) || <ref>[http://dx.doi.org/10.1103/PhysRevLett.85.5138 N. B. Wilding and A. D. Bruce "Freezing by Monte Carlo Phase Switch", Physical Review Letters '''85''' pp. 5138-5141 (2000)]</ref> | |||
|- | |||
| 11.48(11) || <ref name="Miguel"></ref> | |||
|- | |||
| 11.43(17) || <ref>[http://dx.doi.org/10.1063/1.3244562 G. Odriozola "Replica exchange Monte Carlo applied to hard spheres", Journal of Chemical Physics '''131''' 144107 (2009)]</ref> | |||
|- | |||
| 11.550(4) || <ref name="MoirEtAl2021"></ref> | |||
|} | |||
The coexistence [[chemical potential]] has been calculated to be | |||
:{| border="1" | |||
|- | |||
| <math>\mu (k_BT) </math> || Reference | |||
|- | |||
| 15.980(11) || <ref name="Miguel"></ref> | |||
|- | |||
| 16.053(4) || <ref name="MoirEtAl2021"></ref> | |||
|} | |||
The [[Helmholtz energy function]] (in units of <math>Nk_BT</math>) is given by | |||
:{| border="1" | |||
|- | |||
| <math>A_{\mathrm {solid}}</math> || <math>A_{\mathrm {liquid}}</math> || Reference | |||
|- | |||
| 4.887(3) || 3.719(8) || <ref name="Miguel"></ref> | |||
|} | |||
The melting and crystallization process has been studied by Isobe and Krauth <ref>[http://dx.doi.org/10.1063/1.4929529 Masaharu Isobe and Werner Krauth "Hard-sphere melting and crystallization with event-chain Monte Carlo", Journal of Chemical Physics '''143''' 084509 (2015)]</ref>. | |||
==Helmholtz energy function== | |||
Values for the [[Helmholtz energy function]] (<math>A</math>) are given in the following Table: | |||
:{| border="1" | |||
|- | |||
| <math>\rho^*</math> || <math>A/(Nk_BT)</math>|| Reference | |||
|- | |||
| 0.25 || −1.766 <math>\pm</math> 0.002 || Table I <ref name="Schilling"> [http://dx.doi.org/10.1063/1.3274951 T. Schilling and F. Schmid "Computing absolute free energies of disordered structures by molecular simulation", Journal of Chemical Physics '''131''' 231102 (2009)]</ref> | |||
|- | |||
| 0.50 || −0.152 <math>\pm</math> 0.002 || Table I <ref name="Schilling"></ref> | |||
|- | |||
| 0.75 || 1.721 <math>\pm</math> 0.002 || Table I <ref name="Schilling"></ref> | |||
|- | |||
| 1.04086 || 4.959 || Table VI <ref name="VegaNoya"></ref> | |||
|- | |||
| 1.099975 || 5.631 || Table VI <ref name="VegaNoya"></ref> | |||
|- | |||
| 1.150000 || 6.274 || Table VI <ref name="VegaNoya"></ref> | |||
|} | |||
In <ref name="Schilling"></ref> the free energies are given without the ideal gas contribution <math>\ln(\rho^*)-1</math> . Hence, it was added to the free energies in the table. | |||
==Interfacial Helmholtz energy function== | |||
The [[Helmholtz energy function]] of the solid–liquid [[interface]] has been calculated using the [[cleaving method]] giving (Ref. <ref>[http://dx.doi.org/10.1063/1.3514144 Ruslan L. Davidchack "Hard spheres revisited: Accurate calculation of the solid–liquid interfacial free energy", Journal of Chemical Physics '''133''' 234701 (2010)]</ref> Table I): | |||
:{| border="1" | |||
|- | |||
| || [[work]] per unit area/<math>(k_BT/\sigma^2)</math> | |||
|- | |||
| <math>\gamma_{\{100\}}</math> || 0.5820(19) | |||
|- | |||
| <math>\gamma_{\{100\}}</math> || 0.636(11) <ref name="FernandezUCM"></ref> | |||
|- | |||
| <math>\gamma_{\{110\}}</math> || 0.5590(20) | |||
|- | |||
| <math>\gamma_{\{111\}}</math> || 0.5416(31) | |||
|- | |||
| <math>\gamma_{\{120\}}</math> || 0.5669(20) | |||
|} | |||
==Solid structure== | |||
The [http://mathworld.wolfram.com/KeplerConjecture.html Kepler conjecture] states that the optimal packing for three dimensional spheres is either cubic or hexagonal close [[Lattice Structures | packing]], both of which have maximum densities of <math>\pi/(3 \sqrt{2}) \approx 74.048%</math><ref>[http://dx.doi.org/10.1038/26609 Neil J. A. Sloane "Kepler's conjecture confirmed", Nature '''395''' pp. 435-436 (1998)]</ref> | |||
<ref>[https://www.newscientist.com/article/dn26041-proof-confirmed-of-400-year-old-fruit-stacking-problem/ Jacob Aron "Proof confirmed of 400-year-old fruit-stacking problem", New Scientist daily news 12 August (2014)]</ref> | |||
<ref>[http://dx.doi.org/10.1103/PhysRevE.52.3632 C. F. Tejero, M. S. Ripoll, and A. Pérez "Pressure of the hard-sphere solid", Physical Review E '''52''' pp. 3632-3636 (1995)]</ref>. However, for hard spheres at close packing the [[Building up a face centered cubic lattice |face centred cubic]] phase is the more stable | |||
<ref>[http://dx.doi.org/10.1039/a701761h Leslie V. Woodcock "Computation of the free energy for alternative crystal structures of hard spheres", Faraday Discussions '''106''' pp. 325-338 (1997)]</ref>, with a [[Helmholtz energy function]] difference in the [[thermodynamic limit]] between the hexagonal close packed and face centered cubic crystals at close packing of 0.001164(8) <math>Nk_BT</math><ref>[http://dx.doi.org/10.1080/00268976.2014.982736 Eva G. Noya and Noé G. Almarza "Entropy of hard spheres in the close-packing limit", Molecular Physics '''113''' pp. 1061-1068 (2015)]</ref>. Recently evidence has been found for a metastable cI16 phase <ref>[https://doi.org/10.1063/1.5009099 Vadim B. Warshavsky, David M. Ford, and Peter A. Monson "On the mechanical stability of the body-centered cubic phase and the emergence of a metastable cI16 phase in classical hard sphere solids", Journal of Chemical Physics '''148''' 024502 (2018)]</ref> indicating the ''"cI16 is a mechanically stable structure that can spontaneously emerge from a bcc starting point but it is thermodynamically metastable relative to fcc or hcp".'' | |||
*See also: [[Equations of state for crystals of hard spheres]] | |||
==Direct correlation function== | |||
For the [[direct correlation function]] see: | |||
<ref>[http://dx.doi.org/10.1080/00268970701725021 C. F. Tejero and M. López De Haro "Direct correlation function of the hard-sphere fluid", Molecular Physics '''105''' pp. 2999-3004 (2007)]</ref> | |||
<ref>[http://dx.doi.org/10.1080/00268970902784934 Matthew Dennison, Andrew J. Masters, David L. Cheung, and Michael P. Allen "Calculation of direct correlation function for hard particles using a virial expansion", Molecular Physics pp. 375-382 (2009)]</ref> | |||
==Bridge function== | |||
Details of the [[bridge function]] for hard sphere can be found in the following publication | |||
<ref>[http://dx.doi.org/10.1080/00268970210136357 Jiri Kolafa, Stanislav Labik and Anatol Malijevsky "The bridge function of hard spheres by direct inversion of computer simulation data", Molecular Physics '''100''' pp. 2629-2640 (2002)]</ref> | |||
== Equations of state == | == Equations of state == | ||
:''Main article: [[Equations of state for hard spheres]]'' | |||
==Virial coefficients== | |||
:''Main article: [[Hard sphere: virial coefficients]]'' | |||
Hard sphere | == Experimental results == | ||
Pusey and van Megen used a suspension of PMMA particles of radius 305 <math>\pm</math>10 nm, suspended in poly-12-hydroxystearic acid <ref>[http://dx.doi.org/10.1038/320340a0 P. N. Pusey and W. van Megen "Phase behaviour of concentrated suspensions of nearly hard colloidal spheres", Nature '''320''' pp. 340-342 (1986)]</ref> | |||
== | For results obtained from the [http://exploration.grc.nasa.gov/expr2/cdot.html Colloidal Disorder - Order Transition] (CDOT) experiments performed on-board the Space Shuttles ''Columbia'' and ''Discovery'' see Ref. <ref>[http://dx.doi.org/10.1016/S0261-3069(01)00015-2 Z. Chenga, P. M. Chaikina, W. B. Russelb, W. V. Meyerc, J. Zhub, R. B. Rogersc and R. H. Ottewilld, "Phase diagram of hard spheres", Materials & Design '''22''' pp. 529-534 (2001)]</ref> | ||
==Mixtures== | |||
*[[Binary hard-sphere mixtures]] | |||
*[[Multicomponent hard-sphere mixtures]] | |||
== Related systems == | == Related systems == | ||
*[[Quantum hard spheres]] | *[[Quantum hard spheres]] | ||
*[[Dipolar hard spheres]] | *[[Dipolar hard spheres]] | ||
*[[Lattice hard spheres]] | |||
* 1-dimensional case: [[ | Hard spheres in other dimensions: | ||
* 1-dimensional case: [[1-dimensional hard rods | hard rods]]. | |||
* 2-dimensional case: [[Hard disks | hard disks]]. | * 2-dimensional case: [[Hard disks | hard disks]]. | ||
* [[Hard hyperspheres]] | * [[Hard hyperspheres]] | ||
==References== | |||
<references/> | |||
'''Related reading''' | |||
*[http://dx.doi.org/10.1007/978-3-540-78767-9 "Theory and Simulation of Hard-Sphere Fluids and Related Systems", Lecture Notes in Physics '''753/2008''' Springer (2008)] | |||
*[http://dx.doi.org/10.1063/1.3506838 Laura Filion, Michiel Hermes, Ran Ni and Marjolein Dijkstra "Crystal nucleation of hard spheres using molecular dynamics, umbrella sampling, and forward flux sampling: A comparison of simulation techniques", Journal of Chemical Physics '''133''' 244115 (2010)] | |||
==External links== | ==External links== | ||
*[http://www.smac.lps.ens.fr/index.php/Programs_Chapter_2:_Hard_disks_and_spheres Hard disks and spheres] computer code on SMAC-wiki. | *[http://www.smac.lps.ens.fr/index.php/Programs_Chapter_2:_Hard_disks_and_spheres Hard disks and spheres] computer code on SMAC-wiki. | ||
[[Category:Models]] | [[Category:Models]] | ||
[[category: hard sphere]] | [[category: hard sphere]] | ||
Latest revision as of 13:47, 23 May 2023

The hard sphere model (sometimes known as the rigid sphere model) is defined as
where is the intermolecular pair potential between two spheres at a distance , and is the diameter of the sphere. The hard sphere model can be considered to be a special case of the hard ellipsoid model, where each of the semi-axes has the same length, .
First simulations of hard spheres (1954-1957)[edit]
The hard sphere model, along with its two-dimensional manifestation hard disks, was one of the first ever systems studied using computer simulation techniques with a view to understanding the thermodynamics of the liquid and solid phases and their corresponding phase transition [1] [2] [3], much of this work undertaken at the Los Alamos Scientific Laboratory on the world's first electronic digital computer ENIAC [4].
Liquid phase radial distribution function[edit]
The following are a series of plots of the hard sphere radial distribution function [5] shown for different values of the number density . The horizontal axis is in units of where is set to be 1. Click on image of interest to see a larger view.
The value of the radial distribution at contact, , can be used to calculate the pressure via the equation of state (Eq. 1 in [6])
where the second virial coefficient, , is given by
- .
Carnahan and Starling [7] provided the following expression for (Eq. 3 in [6])
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle {\mathrm g}(\sigma^+)= \frac{1-\eta/2}{(1-\eta)^3}}
where Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \eta} is the packing fraction.
Over the years many groups have studied the radial distribution function of the hard sphere model: [8] [9] [10] [11] [12] [13] [14] [15] [16] [17] [18]
Liquid-solid transition[edit]
The hard sphere system undergoes a liquid-solid first order transition [19] [20], sometimes referred to as the Kirkwood-Alder transition [21]. The liquid-solid coexistence densities (Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \rho^* = \rho \sigma^3=6\eta/\pi} ) has been calculated to be
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \rho^*_{\mathrm {solid}}} Reference 1.041(4) 0.943(4) [19] 1.0376 0.9391 [22] 1.0367(10) 0.9387(10) [23] 1.0372 0.9387 [24] 1.0369(33) 0.9375(14) [25] 1.037 0.938 [26] 1.033(3) 0.935(2) [27] 1.03715(9) 0.93890(7) [28]
The coexistence pressure has been calculated to be
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle p (k_BT/\sigma^3) } Reference 11.5727(10) [29] 11.57(10) [23] 11.567 [22] 11.55(11) [30] 11.54(4) [25] 11.50(9) [31] 11.48(11) [27] 11.43(17) [32] 11.550(4) [28]
The coexistence chemical potential has been calculated to be
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \mu (k_BT) } Reference 15.980(11) [27] 16.053(4) [28]
The Helmholtz energy function (in units of Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle Nk_BT} ) is given by
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle A_{\mathrm {solid}}} Reference 4.887(3) 3.719(8) [27]
The melting and crystallization process has been studied by Isobe and Krauth [33].
Helmholtz energy function[edit]
Values for the Helmholtz energy function (Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle A} ) are given in the following Table:
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \rho^*} Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle A/(Nk_BT)} Reference 0.25 −1.766 Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \pm} 0.002 Table I [34] 0.50 −0.152 0.002 Table I [34] 0.75 1.721 Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \pm} 0.002 Table I [34] 1.04086 4.959 Table VI [24] 1.099975 5.631 Table VI [24] 1.150000 6.274 Table VI [24]
In [34] the free energies are given without the ideal gas contribution Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \ln(\rho^*)-1} . Hence, it was added to the free energies in the table.
Interfacial Helmholtz energy function[edit]
The Helmholtz energy function of the solid–liquid interface has been calculated using the cleaving method giving (Ref. [35] Table I):
work per unit area/Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle (k_BT/\sigma^2)} Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \gamma_{\{100\}}} 0.5820(19) 0.636(11) [29] Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \gamma_{\{110\}}} 0.5590(20) Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \gamma_{\{111\}}} 0.5416(31) Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \gamma_{\{120\}}} 0.5669(20)
Solid structure[edit]
The Kepler conjecture states that the optimal packing for three dimensional spheres is either cubic or hexagonal close packing, both of which have maximum densities of Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \pi/(3 \sqrt{2}) \approx 74.048%} [36] [37] [38]. However, for hard spheres at close packing the face centred cubic phase is the more stable [39], with a Helmholtz energy function difference in the thermodynamic limit between the hexagonal close packed and face centered cubic crystals at close packing of 0.001164(8) Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle Nk_BT} [40]. Recently evidence has been found for a metastable cI16 phase [41] indicating the "cI16 is a mechanically stable structure that can spontaneously emerge from a bcc starting point but it is thermodynamically metastable relative to fcc or hcp".
Direct correlation function[edit]
For the direct correlation function see: [42] [43]
Bridge function[edit]
Details of the bridge function for hard sphere can be found in the following publication [44]
Equations of state[edit]
- Main article: Equations of state for hard spheres
Virial coefficients[edit]
- Main article: Hard sphere: virial coefficients
Experimental results[edit]
Pusey and van Megen used a suspension of PMMA particles of radius 305 Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \pm} 10 nm, suspended in poly-12-hydroxystearic acid [45] For results obtained from the Colloidal Disorder - Order Transition (CDOT) experiments performed on-board the Space Shuttles Columbia and Discovery see Ref. [46]
Mixtures[edit]
Related systems[edit]
Hard spheres in other dimensions:
- 1-dimensional case: hard rods.
- 2-dimensional case: hard disks.
- Hard hyperspheres
References[edit]
- ↑ Marshall N. Rosenbluth and Arianna W. Rosenbluth "Further Results on Monte Carlo Equations of State", Journal of Chemical Physics 22 pp. 881-884 (1954)
- ↑ W. W. Wood and J. D. Jacobson "Preliminary Results from a Recalculation of the Monte Carlo Equation of State of Hard Spheres", Journal of Chemical Physics 27 pp. 1207-1208 (1957)
- ↑ B. J. Alder and T. E. Wainwright "Phase Transition for a Hard Sphere System", Journal of Chemical Physics 27 pp. 1208-1209 (1957)
- ↑ The ENIAC Story
- ↑ The total correlation function data was produced using the computer code written by Jiří Kolafa
- ↑ 6.0 6.1 Fu-Ming Tao, Yuhua Song, and E. A. Mason "Derivative of the hard-sphere radial distribution function at contact", Physical Review A 46 pp. 8007-8008 (1992)
- ↑ N. F.Carnahan and K. E.Starling,"Equation of State for Nonattracting Rigid Spheres" Journal of Chemical Physics 51 pp. 635-636 (1969)
- ↑ John G. Kirkwood, Eugene K. Maun, and Berni J. Alder "Radial Distribution Functions and the Equation of State of a Fluid Composed of Rigid Spherical Molecules", Journal of Chemical Physics 18 pp. 1040- (1950)
- ↑ B. R. A. Nijboer and L. Van Hove "Radial Distribution Function of a Gas of Hard Spheres and the Superposition Approximation", Physical Review 85 pp. 777 - 783 (1952)
- ↑ B. J. Alder, S. P. Frankel, and V. A. Lewinson "Radial Distribution Function Calculated by the Monte-Carlo Method for a Hard Sphere Fluid", Journal of Chemical Physics 23 pp. 417- (1955)
- ↑ Francis H. Ree, R. Norris Keeler, and Shaun L. McCarthy "Radial Distribution Function of Hard Spheres", Journal of Chemical Physics 44 pp. 3407- (1966)
- ↑ W. R. Smith and D. Henderson "Analytical representation of the Percus-Yevick hard-sphere radial distribution function", Molecular Physics 19 pp. 411-415 (1970)
- ↑ J. A. Barker and D. Henderson "Monte Carlo values for the radial distribution function of a system of fluid hard spheres", Molecular Physics 21 pp. 187-191 (1971)
- ↑ J. M. Kincaid and J. J. Weis "Radial distribution function of a hard-sphere solid", Molecular Physics 34 pp. 931-938 (1977)
- ↑ S. Bravo Yuste and A. Santos "Radial distribution function for hard spheres", Physical Review A 43 pp. 5418-5423 (1991)
- ↑ Jaeeon Chang and Stanley I. Sandler "A real function representation for the structure of the hard-sphere fluid", Molecular Physics 81 pp. 735-744 (1994)
- ↑ Andrij Trokhymchuk, Ivo Nezbeda and Jan Jirsák "Hard-sphere radial distribution function again", Journal of Chemical Physics 123 024501 (2005)
- ↑ M. López de Haro, A. Santos and S. B. Yuste "On the radial distribution function of a hard-sphere fluid", Journal of Chemical Physics 124 236102 (2006)
- ↑ 19.0 19.1 William G. Hoover and Francis H. Ree "Melting Transition and Communal Entropy for Hard Spheres", Journal of Chemical Physics 49 pp. 3609-3617 (1968)
- ↑ Miguel Robles, Mariano López de Haro and Andrés Santos "Note: Equation of state and the freezing point in the hard-sphere model", Journal of Chemical Physics 140 136101 (2014)
- ↑ Alice P. Gast and William B. Russel "Simple Ordering in Complex Fluids", Physics Today 51 (12) pp. 24-30 (1998)
- ↑ 22.0 22.1 Daan Frenkel and Berend Smit "Understanding Molecular Simulation: From Algorithms to Applications", Second Edition (2002) (ISBN 0-12-267351-4) p. 261.
- ↑ 23.0 23.1 Andrea Fortini and Marjolein Dijkstra "Phase behaviour of hard spheres confined between parallel hard plates: manipulation of colloidal crystal structures by confinement", Journal of Physics: Condensed Matter 18 pp. L371-L378 (2006)
- ↑ 24.0 24.1 24.2 24.3 Carlos Vega and Eva G. Noya "Revisiting the Frenkel-Ladd method to compute the free energy of solids: The Einstein molecule approach", Journal of Chemical Physics 127 154113 (2007)
- ↑ 25.0 25.1 Eva G. Noya, Carlos Vega, and Enrique de Miguel "Determination of the melting point of hard spheres from direct coexistence simulation methods", Journal of Chemical Physics 128 154507 (2008)
- ↑ Ruslan L. Davidchack and Brian B. Laird "Simulation of the hard-sphere crystal–melt interface", Journal of Chemical Physics 108 pp. 9452-9462 (1998)
- ↑ 27.0 27.1 27.2 27.3 Enrique de Miguel "Estimating errors in free energy calculations from thermodynamic integration using fitted data", Journal of Chemical Physics 129 214112 (2008)
- ↑ 28.0 28.1 28.2 Craig Moir, Leo Lue, and Marcus N. Bannerman "Tethered-particle model: The calculation of free energies for hard-sphere systems", Journal of Chemical Physics 155 064504 (2021)
- ↑ 29.0 29.1 L. A. Fernández, V. Martín-Mayor, B. Seoane, and P. Verrocchio "Equilibrium Fluid-Solid Coexistence of Hard Spheres", Physical Review Letters 108 165701 (2012)
- ↑ Robin J. Speedy "Pressure of the metastable hard-sphere fluid", Journal of Physics: Condensed Matter 9 pp. 8591-8599 (1997)
- ↑ N. B. Wilding and A. D. Bruce "Freezing by Monte Carlo Phase Switch", Physical Review Letters 85 pp. 5138-5141 (2000)
- ↑ G. Odriozola "Replica exchange Monte Carlo applied to hard spheres", Journal of Chemical Physics 131 144107 (2009)
- ↑ Masaharu Isobe and Werner Krauth "Hard-sphere melting and crystallization with event-chain Monte Carlo", Journal of Chemical Physics 143 084509 (2015)
- ↑ 34.0 34.1 34.2 34.3 T. Schilling and F. Schmid "Computing absolute free energies of disordered structures by molecular simulation", Journal of Chemical Physics 131 231102 (2009)
- ↑ Ruslan L. Davidchack "Hard spheres revisited: Accurate calculation of the solid–liquid interfacial free energy", Journal of Chemical Physics 133 234701 (2010)
- ↑ Neil J. A. Sloane "Kepler's conjecture confirmed", Nature 395 pp. 435-436 (1998)
- ↑ Jacob Aron "Proof confirmed of 400-year-old fruit-stacking problem", New Scientist daily news 12 August (2014)
- ↑ C. F. Tejero, M. S. Ripoll, and A. Pérez "Pressure of the hard-sphere solid", Physical Review E 52 pp. 3632-3636 (1995)
- ↑ Leslie V. Woodcock "Computation of the free energy for alternative crystal structures of hard spheres", Faraday Discussions 106 pp. 325-338 (1997)
- ↑ Eva G. Noya and Noé G. Almarza "Entropy of hard spheres in the close-packing limit", Molecular Physics 113 pp. 1061-1068 (2015)
- ↑ Vadim B. Warshavsky, David M. Ford, and Peter A. Monson "On the mechanical stability of the body-centered cubic phase and the emergence of a metastable cI16 phase in classical hard sphere solids", Journal of Chemical Physics 148 024502 (2018)
- ↑ C. F. Tejero and M. López De Haro "Direct correlation function of the hard-sphere fluid", Molecular Physics 105 pp. 2999-3004 (2007)
- ↑ Matthew Dennison, Andrew J. Masters, David L. Cheung, and Michael P. Allen "Calculation of direct correlation function for hard particles using a virial expansion", Molecular Physics pp. 375-382 (2009)
- ↑ Jiri Kolafa, Stanislav Labik and Anatol Malijevsky "The bridge function of hard spheres by direct inversion of computer simulation data", Molecular Physics 100 pp. 2629-2640 (2002)
- ↑ P. N. Pusey and W. van Megen "Phase behaviour of concentrated suspensions of nearly hard colloidal spheres", Nature 320 pp. 340-342 (1986)
- ↑ Z. Chenga, P. M. Chaikina, W. B. Russelb, W. V. Meyerc, J. Zhub, R. B. Rogersc and R. H. Ottewilld, "Phase diagram of hard spheres", Materials & Design 22 pp. 529-534 (2001)
Related reading
- "Theory and Simulation of Hard-Sphere Fluids and Related Systems", Lecture Notes in Physics 753/2008 Springer (2008)
- Laura Filion, Michiel Hermes, Ran Ni and Marjolein Dijkstra "Crystal nucleation of hard spheres using molecular dynamics, umbrella sampling, and forward flux sampling: A comparison of simulation techniques", Journal of Chemical Physics 133 244115 (2010)
External links[edit]
- Hard disks and spheres computer code on SMAC-wiki.