SPC/E model of water: Difference between revisions
Jump to navigation
Jump to search
Carl McBride (talk | contribs) m (Added parameters + a new reference) |
m (slight reparametrization) |
||
| Line 1: | Line 1: | ||
{{Stub-water}} | {{Stub-water}} | ||
The extended simple point charge model, '''SPC/E''' is a reparameterisation of the [[SPC]] model of [[water]]. | The extended simple point charge model, '''SPC/E''' is a slight reparameterisation of the [[SPC]] model of [[water]], with a modified value for <math>q_{\mathrm{O}}</math>. | ||
The molecule is modelled as | The molecule is modelled as | ||
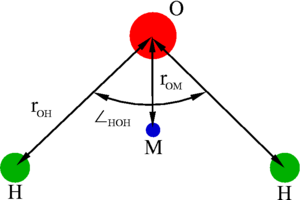
a rigid isosceles triangle, having charges situated on each of the three atoms. Apart from Coulombic interactions, the molecules interact via long-range [[Lennard-Jones model | Lennard-Jones]] sites, situated on the oxygen atoms. The parameters are as follows: | a rigid isosceles triangle, having charges situated on each of the three atoms. Apart from Coulombic interactions, the molecules interact via long-range [[Lennard-Jones model | Lennard-Jones]] sites, situated on the oxygen atoms. The parameters are as follows: | ||
Revision as of 14:51, 23 May 2008
The extended simple point charge model, SPC/E is a slight reparameterisation of the SPC model of water, with a modified value for . The molecule is modelled as a rigid isosceles triangle, having charges situated on each of the three atoms. Apart from Coulombic interactions, the molecules interact via long-range Lennard-Jones sites, situated on the oxygen atoms. The parameters are as follows:
| parameter | value |
| kJ mol-1 | |
| (charge neutrality) | |
| (charge sits on oxygen) |
The SPC/E model has a dipole moment of 2.351 D. (Ref. 1 Table I),
References
- H. J. C. Berendsen, J. R. Grigera, and T. P. Straatsma "The missing term in effective pair potentials", Journal of Physical Chemistry 91 pp. 6269 - 6271 (1987)
- Swaroop Chatterjee, Pablo G. Debenedetti, Frank H. Stillinger, and Ruth M. Lynden-Bell "A computational investigation of thermodynamics, structure, dynamics and solvation behavior in modified water models", Journal of Chemical Physics 128 124511 (2008)













