Argon: Difference between revisions
(Ar mass added) |
Carl McBride (talk | contribs) m (Added a recent publication) |
||
| (41 intermediate revisions by 4 users not shown) | |||
| Line 1: | Line 1: | ||
'''Argon''' (Ar) has long been a popular choice for [[Computer simulation techniques |computer simulations]] of simple liquids. Some of the first computer simulations of liquid argon were those of Wood and Parker in 1957 <ref>[http://dx.doi.org/10.1063/1.1743822 W. W. Wood and F. R. Parker "Monte Carlo Equation of State of Molecules Interacting with the Lennard‐Jones Potential. I. A Supercritical Isotherm at about Twice the Critical Temperature", Journal of Chemical Physics '''27''' pp. 720- (1957)]</ref> and of Rahman in 1964 <ref name="Rahman">[http://dx.doi.org/10.1103/PhysRev.136.A405 A. Rahman "Correlations in the Motion of Atoms in Liquid Argon", Physical Review '''136''' pp. A405–A411 (1964)]</ref>. Sadus and Prausnitz have shown that three-body repulsion makes a significant contribution to [[Idealised models#Three-body potentials|three-body interactions]] in the liquid phase | |||
[[ | <ref>[http://dx.doi.org/10.1063/1.471172 Richard J. Sadus and J. M. Prausnitz "Three-body interactions in fluids from molecular simulation: Vapor–liquid phase coexistence of argon", Journal of Chemical Physics '''104''' pp. 4784-4787 (1996)]</ref> | ||
(for use of the [[Axilrod-Teller interaction]] see | |||
<ref>[http://dx.doi.org/10.1103/PhysRevA.45.3659 Phil Attard "Pair-hypernetted-chain closure for three-body potentials: Results for argon with the Axilrod-Teller triple-dipole potential", Physical Review A '''45''' pp. 3659-3669 (1992)]</ref> | |||
<ref>[http://dx.doi.org/10.1103/PhysRevE.55.2707 J. A. Anta, E. Lomba and M. Lombardero "Influence of three-body forces on the gas-liquid coexistence of simple fluids: The phase equilibrium of argon", Physical Review E '''55''' pp. 2707-2712 (1997)]</ref>). | |||
However, the generic [[Lennard-Jones model]] has been frequently used due to its simplicity; some parameters are quoted in the next section. A specific interatomic potential for Ar has been proposed by Aziz | |||
<ref>[http://dx.doi.org/10.1063/1.466051 Ronald A. Aziz "A highly accurate interatomic potential for argon", Journal of Chemical Physics '''99''' p. 4518 (1993)]</ref>. | |||
==Thermophysical properties (experimental) == | |||
{| border="1" | |||
|- | |||
| Property <ref>[http://www.webelements.com/argon/physics.html Physical properties of Argon on webelements] </ref> || Temperature || Pressure | |||
|- | |||
| Triple point || 83.8058 K || 69 kPa | |||
|- | |||
| Critical point || 150.87 K || 4.898 MPa | |||
|- | |||
| Melting point || 83.80 K || | |||
|- | |||
| Boiling point ||87.30 K || | |||
|} | |||
==Models== | |||
====Lennard-Jones parameters==== | |||
[[Image:Lennard-Jones.png|thumb| The Lennard-Jones model for argon (Rowley, Nicholson and Parsonage parameters).]] | |||
A selection of parameters for the [[Lennard-Jones model]] for liquid argon are listed in the following table: | |||
{| border="1" | |||
|- | |||
| Authors || <math>\epsilon/k_B</math> (K) || <math>\sigma</math> (nm)|| Reference (year) | |||
|- | |||
| Rahman || 120 || 0.34 || <ref name="Rahman"></ref> (1964) | |||
|- | |||
|Barker, Fisher and Watts ||142.095 || 0.33605 || <ref>[http://dx.doi.org/10.1080/00268977100101821 J. A. Barker, R. A. Fisher and R. O. Watts "Liquid argon: Monte carlo and molecular dynamics calculations", Molecular Physics '''21''' pp. 657-673 (1971)]</ref> (1971) | |||
|- | |||
| Rowley, Nicholson and Parsonage || 119.8 || 0.3405 || <ref>[http://dx.doi.org/10.1016/0021-9991(75)90042-X L. A. Rowley, D. Nicholson and N. G. Parsonage "Monte Carlo grand canonical ensemble calculation in a gas-liquid transition region for 12-6 Argon", Journal of Computational Physics '''17''' pp. 401-414 (1975)]</ref> (1975) | |||
|- | |||
| White || 125.7 || 0.3345 || <ref>[http://dx.doi.org/10.1063/1.479848 John A. White "Lennard-Jones as a model for argon and test of extended renormalization group calculations", Journal of Chemical Physics '''111''' pp. 9352-9356 (1999)]</ref> parameter set #4 (1999) | |||
|} | |||
====Buckingham potential==== | |||
The [[Buckingham potential]] for argon is given by (Eq. 27 <ref>[http://dx.doi.org/10.1098/rspa.1938.0173 R. A. Buckingham "The Classical Equation of State of Gaseous Helium, Neon and Argon", Proceedings of the Royal Society of London. Series A, Mathematical and Physical Sciences '''168''' pp. 264-283 (1938)]</ref>): | |||
:<math>E(r) = 1.69 \times 10^{-8} e^{-r/0.273} -102 \times 10^{-12} r^{-6} </math> | |||
where <math>E</math> is in ergs ( 10<sup>−7</sup> J) and <math>r</math> in Å. | |||
====BBMS potential==== | |||
The Bobetic-Barker-Maitland-Smith potential <ref>[http://dx.doi.org/10.1103/PhysRevB.2.4169 M. V. Bobetic and J. A. Barker "Lattice Dynamics with Three-Body Forces: Argon", Physical Review B '''2''' 4169-4175 (1970)]</ref> <ref>[http://dx.doi.org/10.1080/00268977100103181 G. C. Maitland and E. B. Smith "The intermolecular pair potential of argon", Molecular Physics '''22''' pp. 861-868 (1971)]</ref>. | |||
====Dymond-Alder pair potential==== | |||
<ref>[http://dx.doi.org/10.1063/1.1671724 J. H. Dymond and B. J. Alder "Pair Potential for Argon", Journal of Chemical Physics '''51''' pp. 309-320 (1969)]</ref> | |||
==Radial distribution function== | |||
[[Radial distribution function]] <ref>[http://dx.doi.org/10.1103/PhysRevA.7.2130 J. L. Yarnell, M. J. Katz, R. G. Wenzel and S. H. Koenig "Structure Factor and Radial Distribution Function for Liquid Argon at 85°K", Physical Review A '''7''' pp. 2130-2144 (1973)]</ref> | |||
==Nucleation== | |||
[[Supercooling and nucleation | Nucleation]]<ref>[http://dx.doi.org/10.1063/1.3474945 Matthew J. McGrath, Julius N. Ghogomu, Narcisse T. Tsona, J. Ilja Siepmann, Bin Chen, Ismo Napari1, and Hanna Vehkamäki "Vapor-liquid nucleation of argon: Exploration of various intermolecular potentials", Journal of Chemical Physics '''133''' 084106 (2010)]</ref> | |||
==Quantum simulations== | |||
<ref>[http://dx.doi.org/10.1080/00268978900100811 J. R. Melrose and K. Singer "An investigation of supercooled Lennard-Jones argon by quantum mechanical and classical Monte Carlo simulation", Molecular Physics '''66''' 1203-1214 (1989)]</ref> | |||
==Virial equation of state== | |||
[[Virial equation of state]] <ref>[http://dx.doi.org/10.1063/1.3627151 Benjamin Jäger, Robert Hellmann, Eckard Bich, and Eckhard Vogel "Ab initio virial equation of state for argon using a new nonadditive three-body potential", Journal of Chemical Physics '''135''' 084308 (2011)]</ref> | |||
==Viscosity== | |||
[[Viscosity]] <ref>[http://dx.doi.org/10.1063/1.4704930 S. V. Lishchuk "Role of three-body interactions in formation of bulk viscosity in liquid argon", Journal of Chemical Physics '''136''' 164501 (2012)]</ref>. | |||
==Heat capacity== | |||
[[Heat capacity]] <ref>[http://dx.doi.org/10.1080/00268976.2012.655795 Ali Morsali, S. Ali Beyramabadi, S. Hooman Vahidi & Maryam Ghorbani "A molecular dynamics study on the role of attractive and repulsive forces in excess heat capacity at constant volume of dense fluids", Molecular Physics '''110''' pp. 483-490 (2012)]</ref>. | |||
==Surface tension== | |||
[[Surface tension]] <ref>[http://dx.doi.org/10.1063/1.4885351 Florent Goujon, Patrice Malfreyt and Dominic J. Tildesley "The gas-liquid surface tension of argon: A reconciliation between experiment and simulation", Journal of Chemical Physics '''140''' 244710 (2014)]</ref>. | |||
==Liquid-vapour coexistence== | |||
<ref>[https://doi.org/10.1063/1.5025726 Himanshu Goel, Sanliang Ling, Breanna Nicole Ellis, Anna Taconi, Ben Slater, and Neeraj Rai "Predicting vapor liquid equilibria using density functional theory: A case study of argon", Journal of Chemical Physics 148, 224501 (2018)]</ref>. | |||
==References== | |||
<references/> | |||
'''Related material''' | |||
*[http://dx.doi.org/10.1080/00268976400100611 H. C. Longuet-Higgins and B. Widom "A rigid sphere model for the melting of argon", Molecular Physics '''8''' pp. 549-556 (1964)] | |||
*[http://dx.doi.org/10.1080/00268976800100721 D. Henderson and J. A. Barker "On the solidification of argon", Molecular Physics '''14''' pp. 587-589 (1968)] | |||
*[http://dx.doi.org/10.1103/PhysRevA.5.2238 F. Lado "Numerical Calculation of the Density Autocorrelation Function for Liquid Argon", Physical Review A '''5''' pp. 2238-2244 (1972)] | |||
*[http://dx.doi.org/10.1016/j.molliq.2009.09.009 Ali Asghar Davoodi and Farzaneh Feyzi "A new approach for long range corrections in molecular dynamics simulation with application to calculation of argon properties", Journal of Molecular Liquids '''150''' pp. 33-38 (2009)] | |||
*[http://dx.doi.org/10.1063/1.4740465 Jonas Wiebke, Elke Pahl, and Peter Schwerdtfeger "Sensitivity of the thermal and acoustic virial coefficients of argon to the argon interaction potential", Journal of Chemical Physics '''137''' 064702 (2012)] | |||
*[http://dx.doi.org/10.1080/00268976.2013.798696 C. Fan, D.D. Do, D. Nicholson and E. Ustinov "Chemical potential, Helmholtz free energy and entropy of argon with kinetic Monte Carlo simulation", Molecular Physics '''112''' pp. 60-73 (2014)] | |||
*[https://doi.org/10.1016/j.molliq.2017.01.079 Nikolay P. Malomuzh, and Konstantin S. Shakun "Specific properties of argon-like liquids near their spinodals", Journal of Molecular Liquids '''235''' 155-162 (2017)] | |||
*[http://dx.doi.org/10.1080/00268976.2016.1246760 Gábor Rutkai, Monika Thol, Roland Span and Jadran Vrabec "How well does the Lennard-Jones potential represent the thermodynamic properties of noble gases?", Molecular Physics '''115''' pp. 1104-1121 (2017)] | |||
*[http://dx.doi.org/10.1063/1.4991012 Maryna Vlasiuk and Richard J. Sadus "Ab initio interatomic potentials and the thermodynamic properties of fluids", Journal of Chemical Physics '''147''' 024505 (2017)] | |||
[[category: models]] | [[category: models]] | ||
{{Numeric}} | |||
Latest revision as of 11:55, 18 June 2018
Argon (Ar) has long been a popular choice for computer simulations of simple liquids. Some of the first computer simulations of liquid argon were those of Wood and Parker in 1957 [1] and of Rahman in 1964 [2]. Sadus and Prausnitz have shown that three-body repulsion makes a significant contribution to three-body interactions in the liquid phase [3] (for use of the Axilrod-Teller interaction see [4] [5]). However, the generic Lennard-Jones model has been frequently used due to its simplicity; some parameters are quoted in the next section. A specific interatomic potential for Ar has been proposed by Aziz [6].
Thermophysical properties (experimental)[edit]
| Property [7] | Temperature | Pressure |
| Triple point | 83.8058 K | 69 kPa |
| Critical point | 150.87 K | 4.898 MPa |
| Melting point | 83.80 K | |
| Boiling point | 87.30 K |
Models[edit]
Lennard-Jones parameters[edit]
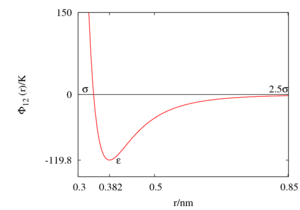
A selection of parameters for the Lennard-Jones model for liquid argon are listed in the following table:
| Authors | (K) | (nm) | Reference (year) |
| Rahman | 120 | 0.34 | [2] (1964) |
| Barker, Fisher and Watts | 142.095 | 0.33605 | [8] (1971) |
| Rowley, Nicholson and Parsonage | 119.8 | 0.3405 | [9] (1975) |
| White | 125.7 | 0.3345 | [10] parameter set #4 (1999) |
Buckingham potential[edit]
The Buckingham potential for argon is given by (Eq. 27 [11]):
where is in ergs ( 10−7 J) and in Å.
BBMS potential[edit]
The Bobetic-Barker-Maitland-Smith potential [12] [13].
Dymond-Alder pair potential[edit]
Radial distribution function[edit]
Radial distribution function [15]
Nucleation[edit]
Quantum simulations[edit]
Virial equation of state[edit]
Viscosity[edit]
Heat capacity[edit]
Surface tension[edit]
Liquid-vapour coexistence[edit]
[22].
References[edit]
- ↑ W. W. Wood and F. R. Parker "Monte Carlo Equation of State of Molecules Interacting with the Lennard‐Jones Potential. I. A Supercritical Isotherm at about Twice the Critical Temperature", Journal of Chemical Physics 27 pp. 720- (1957)
- ↑ 2.0 2.1 A. Rahman "Correlations in the Motion of Atoms in Liquid Argon", Physical Review 136 pp. A405–A411 (1964)
- ↑ Richard J. Sadus and J. M. Prausnitz "Three-body interactions in fluids from molecular simulation: Vapor–liquid phase coexistence of argon", Journal of Chemical Physics 104 pp. 4784-4787 (1996)
- ↑ Phil Attard "Pair-hypernetted-chain closure for three-body potentials: Results for argon with the Axilrod-Teller triple-dipole potential", Physical Review A 45 pp. 3659-3669 (1992)
- ↑ J. A. Anta, E. Lomba and M. Lombardero "Influence of three-body forces on the gas-liquid coexistence of simple fluids: The phase equilibrium of argon", Physical Review E 55 pp. 2707-2712 (1997)
- ↑ Ronald A. Aziz "A highly accurate interatomic potential for argon", Journal of Chemical Physics 99 p. 4518 (1993)
- ↑ Physical properties of Argon on webelements
- ↑ J. A. Barker, R. A. Fisher and R. O. Watts "Liquid argon: Monte carlo and molecular dynamics calculations", Molecular Physics 21 pp. 657-673 (1971)
- ↑ L. A. Rowley, D. Nicholson and N. G. Parsonage "Monte Carlo grand canonical ensemble calculation in a gas-liquid transition region for 12-6 Argon", Journal of Computational Physics 17 pp. 401-414 (1975)
- ↑ John A. White "Lennard-Jones as a model for argon and test of extended renormalization group calculations", Journal of Chemical Physics 111 pp. 9352-9356 (1999)
- ↑ R. A. Buckingham "The Classical Equation of State of Gaseous Helium, Neon and Argon", Proceedings of the Royal Society of London. Series A, Mathematical and Physical Sciences 168 pp. 264-283 (1938)
- ↑ M. V. Bobetic and J. A. Barker "Lattice Dynamics with Three-Body Forces: Argon", Physical Review B 2 4169-4175 (1970)
- ↑ G. C. Maitland and E. B. Smith "The intermolecular pair potential of argon", Molecular Physics 22 pp. 861-868 (1971)
- ↑ J. H. Dymond and B. J. Alder "Pair Potential for Argon", Journal of Chemical Physics 51 pp. 309-320 (1969)
- ↑ J. L. Yarnell, M. J. Katz, R. G. Wenzel and S. H. Koenig "Structure Factor and Radial Distribution Function for Liquid Argon at 85°K", Physical Review A 7 pp. 2130-2144 (1973)
- ↑ Matthew J. McGrath, Julius N. Ghogomu, Narcisse T. Tsona, J. Ilja Siepmann, Bin Chen, Ismo Napari1, and Hanna Vehkamäki "Vapor-liquid nucleation of argon: Exploration of various intermolecular potentials", Journal of Chemical Physics 133 084106 (2010)
- ↑ J. R. Melrose and K. Singer "An investigation of supercooled Lennard-Jones argon by quantum mechanical and classical Monte Carlo simulation", Molecular Physics 66 1203-1214 (1989)
- ↑ Benjamin Jäger, Robert Hellmann, Eckard Bich, and Eckhard Vogel "Ab initio virial equation of state for argon using a new nonadditive three-body potential", Journal of Chemical Physics 135 084308 (2011)
- ↑ S. V. Lishchuk "Role of three-body interactions in formation of bulk viscosity in liquid argon", Journal of Chemical Physics 136 164501 (2012)
- ↑ Ali Morsali, S. Ali Beyramabadi, S. Hooman Vahidi & Maryam Ghorbani "A molecular dynamics study on the role of attractive and repulsive forces in excess heat capacity at constant volume of dense fluids", Molecular Physics 110 pp. 483-490 (2012)
- ↑ Florent Goujon, Patrice Malfreyt and Dominic J. Tildesley "The gas-liquid surface tension of argon: A reconciliation between experiment and simulation", Journal of Chemical Physics 140 244710 (2014)
- ↑ Himanshu Goel, Sanliang Ling, Breanna Nicole Ellis, Anna Taconi, Ben Slater, and Neeraj Rai "Predicting vapor liquid equilibria using density functional theory: A case study of argon", Journal of Chemical Physics 148, 224501 (2018)
Related material
- H. C. Longuet-Higgins and B. Widom "A rigid sphere model for the melting of argon", Molecular Physics 8 pp. 549-556 (1964)
- D. Henderson and J. A. Barker "On the solidification of argon", Molecular Physics 14 pp. 587-589 (1968)
- F. Lado "Numerical Calculation of the Density Autocorrelation Function for Liquid Argon", Physical Review A 5 pp. 2238-2244 (1972)
- Ali Asghar Davoodi and Farzaneh Feyzi "A new approach for long range corrections in molecular dynamics simulation with application to calculation of argon properties", Journal of Molecular Liquids 150 pp. 33-38 (2009)
- Jonas Wiebke, Elke Pahl, and Peter Schwerdtfeger "Sensitivity of the thermal and acoustic virial coefficients of argon to the argon interaction potential", Journal of Chemical Physics 137 064702 (2012)
- C. Fan, D.D. Do, D. Nicholson and E. Ustinov "Chemical potential, Helmholtz free energy and entropy of argon with kinetic Monte Carlo simulation", Molecular Physics 112 pp. 60-73 (2014)
- Nikolay P. Malomuzh, and Konstantin S. Shakun "Specific properties of argon-like liquids near their spinodals", Journal of Molecular Liquids 235 155-162 (2017)
- Gábor Rutkai, Monika Thol, Roland Span and Jadran Vrabec "How well does the Lennard-Jones potential represent the thermodynamic properties of noble gases?", Molecular Physics 115 pp. 1104-1121 (2017)
- Maryna Vlasiuk and Richard J. Sadus "Ab initio interatomic potentials and the thermodynamic properties of fluids", Journal of Chemical Physics 147 024505 (2017)




