Carnahan-Starling equation of state
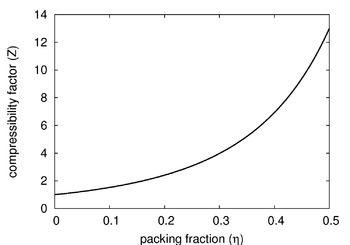
The Carnahan-Starling equation of state is an approximate (but quite good) equation of state for the fluid phase of the hard sphere model in three dimensions. It is given by (Ref [1] Eqn. 10).
where:
- is the compressibility factor
- is the pressure
- is the volume
- is the number of particles
- is the Boltzmann constant
- is the absolute temperature
- is the packing fraction:
- is the hard sphere diameter.
The Carnahan-Starling equation of state is not applicable for packing fractions greater than 0.55 [2].
Virial expansion[edit]
It is interesting to compare the virial coefficients of the Carnahan-Starling equation of state (Eq. 7 of [1]) with the hard sphere virial coefficients in three dimensions (exact up to Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle B_4} , and those of Clisby and McCoy [3]):
| Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle n} | Clisby and McCoy | Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle B_n=n^2+n-2} |
| 2 | 4 | 4 |
| 3 | 10 | 10 |
| 4 | 18.3647684 | 18 |
| 5 | 28.22451(26) | 28 |
| 6 | 39.81515(93) | 40 |
| 7 | 53.3444(37) | 54 |
| 8 | 68.538(18) | 70 |
| 9 | 85.813(85) | 88 |
| 10 | 105.78(39) | 108 |
Thermodynamic expressions[edit]
From the Carnahan-Starling equation for the fluid phase the following thermodynamic expressions can be derived (Ref [4] Eqs. 2.6, 2.7 and 2.8)
Pressure (compressibility):
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \frac{p^{CS}V}{N k_B T } = \frac{1+ \eta + \eta^2 - \eta^3}{(1-\eta)^3}}
Configurational chemical potential:
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \frac{ \overline{\mu }^{CS}}{k_B T} = \frac{8\eta -9 \eta^2 + 3\eta^3}{(1-\eta)^3}}
Isothermal compressibility:
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \chi_T -1 = \frac{1}{k_BT} \left.\frac{\partial P^{CS}}{\partial \rho}\right\vert_{T} -1 = \frac{8\eta -2 \eta^2 }{(1-\eta)^4}}
where Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \eta} is the packing fraction.
Configurational Helmholtz energy function:
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle \frac{ A_{ex}^{CS}}{N k_B T} = \frac{4 \eta - 3 \eta^2 }{(1-\eta)^2}}
The 'Percus-Yevick' derivation[edit]
It is interesting to note (Ref [5] Eq. 6) that one can arrive at the Carnahan-Starling equation of state by adding two thirds of the exact solution of the Percus Yevick integral equation for hard spheres via the compressibility route, to one third via the pressure route, i.e.
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle Z = \frac{ p V}{N k_B T} = \frac{2}{3} \left[ \frac{(1+\eta+\eta^2)}{(1-\eta)^3} \right] + \frac{1}{3} \left[ \frac{(1+2\eta+3\eta^2)}{(1-\eta)^2} \right] = \frac{ 1 + \eta + \eta^2 - \eta^3 }{(1-\eta)^3 }}
The reason for this seems to be a slight mystery (see discussion in Ref. [6] ).
Kolafa correction[edit]
Jiri Kolafa produced a slight correction to the C-S EOS which results in improved accuracy [7]:
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle Z = \frac{ 1 + \eta + \eta^2 - \frac{2}{3}(1+\eta) \eta^3 }{(1-\eta)^3 }. }
Liu correction[edit]
Hongqin Liu proposed a correction to the C-S EOS which improved accuracy by almost two order of magnitude [8]:
- Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle Z = \frac{ 1 + \eta + \eta^2 - \frac{8}{13}\eta^3 - \eta^4 + \frac{1}{2}\eta^5 }{(1-\eta)^3 }. }
See also[edit]
References[edit]
- ↑ 1.0 1.1 N. F. Carnahan and K. E. Starling,"Equation of State for Nonattracting Rigid Spheres" Journal of Chemical Physics 51 pp. 635-636 (1969)
- ↑ Hongqin Liu "A very accurate hard sphere equation of state over the entire stable and metstable region", arXiv:cond-mat/0605392 (2006)
- ↑ Nathan Clisby and Barry M. McCoy "Ninth and Tenth Order Virial Coefficients for Hard Spheres in D Dimensions", Journal of Statistical Physics 122 pp. 15-57 (2006)
- ↑ Lloyd L. Lee "An accurate integral equation theory for hard spheres: Role of the zero-separation theorems in the closure relation", Journal of Chemical Physics 103 pp. 9388-9396 (1995)
- ↑ G. A. Mansoori, N. F. Carnahan, K. E. Starling, and T. W. Leland, Jr. "Equilibrium Thermodynamic Properties of the Mixture of Hard Spheres", Journal of Chemical Physics 54 pp. 1523-1525 (1971)
- ↑ Yuhua Song, E. A. Mason, and Richard M. Stratt "Why does the Carnahan-Starling equation work so well?", Journal of Physical Chemistry 93 pp. 6916-6919 (1989)
- ↑ Miguel Robles, Mariano López de Haro and Andrés Santos "Note: Equation of state and the freezing point in the hard-sphere model", Journal of Chemical Physics 140 136101 (2014)
- ↑ Hongqin Liu "Carnahan Starling type equations of state for stable hard disk and hard sphere fluids", arXiv:2010.14357









