Lennard-Jones model
The Lennard-Jones potential was developed by Sir John Edward Lennard-Jones.
Lennard-Jones potential
The Lennard-Jones potential is given by:
where:
- is the intermolecular pair potential between two particles at a distance r;
- : diameter (length);
- : well depth (energy)
Reduced units:
- Density, , where (number of particles divided by the volume .)
- Temperature; , where is the absolute temperature and is the Boltzmann constant
Argon
The Lennard-Jones parameters for argon are 119.8 K and 0.3405 nm. (Ref. ?)
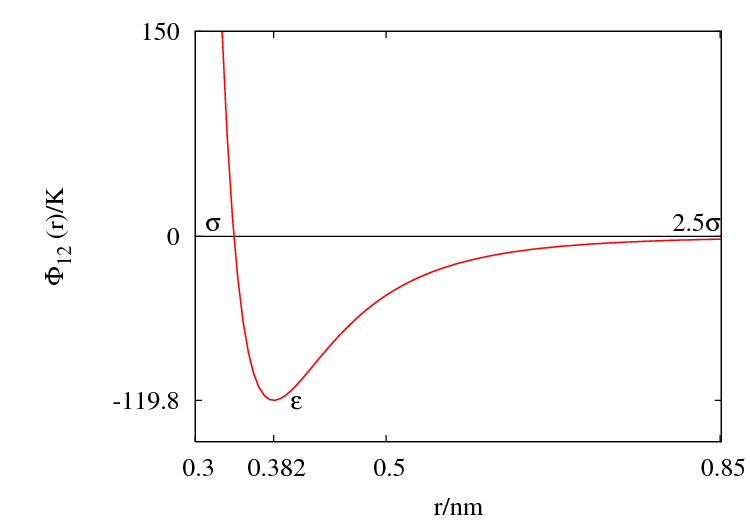
This figure was produced using gnuplot with the command:
plot (4*120*((0.34/x)**12-(0.34/x)**6))
Features
Special points:
- Minimum value of at ;
Critical point
The location of the critical point is
at a reduced density of
- .
Caillol (Ref. 3) reports and .
Triple point
The location of the triple point as found by Mastny and de Pablo (Ref. 2) is
Approximations in simulation: truncation and shifting
The Lennard-Jones model is often used with a cutoff radius of . See Mastny and de Pablo (Ref. 2) fa an analysis of the effect of this cutoff on the melting line.
Related potential models
It is relatively common the use of potential functions given by:
with and being positive integer numbers and , and is chosen to get the minimum value of being
These forms are usually referred to as m-n Lennard-Jones Potential.
The 9-3 Lennard-Jones interaction potential is often use to model the interaction between the atoms/molecules of a fluid and a continuous solid wall. In (9-3 Lennard-Jones potential) a justification of this use is presented.
Other dimensions
- 1-dimensional case: Lennard-Jones rods.
- 2-dimensional case: Lennard-Jones disks.
See also
- Phase diagram of the Lennard-Jones model
- Lennard-Jones model: virial coefficients
- Lennard-Jones equation of state
References
- J. E. Lennard-Jones, "Cohesion", Proceedings of the Physical Society, 43 pp. 461-482 (1931)
- Ethan A. Mastny and Juan J. de Pablo "Melting line of the Lennard-Jones system, infinite size, and full potential", Journal of Chemical Physics 127 104504 (2007)
- J. M. Caillol " Critical-point of the Lennard-Jones fluid: A finite-size scaling study", Journal of Chemical Physics 109 4885-4893(1008)
![{\displaystyle \Phi (r)=4\epsilon \left[\left({\frac {\sigma }{r}}\right)^{12}-\left({\frac {\sigma }{r}}\right)^{6}\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/b0ecd4ec335b5149b5f30ba2d02a6049d0407207)





















![{\displaystyle \Phi (r)=c_{m,n}\epsilon \left[\left({\frac {\sigma }{r}}\right)^{m}-\left({\frac {\sigma }{r}}\right)^{n}\right].}](https://wikimedia.org/api/rest_v1/media/math/render/svg/58317a77137d2a636f30e2a4a48c2ba87b516598)




