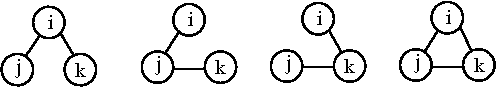Cluster integrals
In an ideal gas there are no intermolecular interactions. However, in an imperfect or real gas, this is not so, and the second virial coefficient is other than zero. Mayer and Mayer developed a theoretical treatment of the virial coefficients in terms of cluster integrals.
The simplest cluster is that consisting of a single molecule, not bound to any other. A cluster of three specified identical molecules, i, j and k may be formed in any of four ways:
The first three cluster integrals are (Eq. 13.6 in [1])
Ref. 1 Eq. 13.7:
and Ref. 1 Eq. 13.8:
using the Mayer f-function notation.
Irreducible clusters[edit]
Irreducible clusters are denoted by
note .
note
note
Hellmann and Bich diagrams[edit]
Hellmann and Bich have rederived the virial equation of state from the grand canonical partition function without restricting themselves to pairwise intermolecular pair potentials [2]. This leads to expressions for the virial coefficients that, for and beyond, require the evaluation of far fewer diagrams when compared to the original diagrams of Mayer or to the reformulation of Ree and Hoover [3].
See also[edit]
References[edit]
- ↑ Joseph Edward Mayer and Maria Goeppert Mayer "Statistical Mechanics" John Wiley and Sons (1940) Chapter 13
- ↑ Robert Hellmann and Eckard Bich "A systematic formulation of the virial expansion for nonadditive interaction potentials", Journal of Chemical Physics 135 084117 (2011)
- ↑ Francis H. Ree and William G. Hoover "Reformulation of the Virial Series for Classical Fluids", Journal of Chemical Physics 41 1635 (1964)
- Related reading