Fused hard sphere chains: Difference between revisions
Jump to navigation
Jump to search
| Line 36: | Line 36: | ||
The Waziri and Hamad [[Equations of state | equation of state]] for fused hard sphere chain fluids is given by | The Waziri and Hamad [[Equations of state | equation of state]] for fused hard sphere chain fluids is given by | ||
:<math>Z_{FHSC} = 1 + 4m_{\mathrm{eff}}P^{*} + \frac{3}{4}m_{\mathrm{eff}}P^{*}\ln\left[\frac{3+P^{*}}{3+25P^{*}}\right] + \frac{216(m_{\mathrm{eff}} - 1)P^{*}}{(3+P^{*})(3+25P^{*})\{16+3\ln[(3+P^{*})/(3+25P^{*})]\}}</math> | :<math>Z_{\mathrm{FHSC}} = 1 + 4m_{\mathrm{eff}}P^{*} + \frac{3}{4}m_{\mathrm{eff}}P^{*}\ln\left[\frac{3+P^{*}}{3+25P^{*}}\right] + \frac{216(m_{\mathrm{eff}} - 1)P^{*}}{(3+P^{*})(3+25P^{*})\{16+3\ln[(3+P^{*})/(3+25P^{*})]\}}</math> | ||
where | where | ||
Revision as of 11:35, 18 December 2018
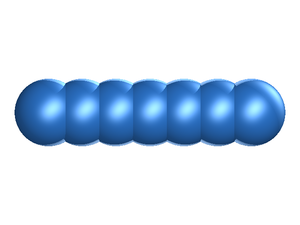
In the fused hard sphere chain model the molecule is built up form a string of overlapping hard sphere sites, each of diameter .
An effective number of monomers can be applied to the fused hard sphere chain model by using the relarion (Ref. 4 Eq. 2.18)
where m is the number of monomer units in the model, and is the reduced bond length.
The volume of the fused hard sphere chain is given by (Ref. 5 Eq. 13)
where is the minimal bond angle, and the surface area is given by (Ref. 5 Eq. 12)
Equation of state
The Vörtler and Nezbeda equation of state is given by
where
and
The Waziri and Hamad equation of state for fused hard sphere chain fluids is given by
where
- Horst L. Vörtler and I. Nezbeda "Volume-explicit equation of state and excess volume of mixing of fused hard sphere fluids", Berichte der Bunsen-Gesellschaft 94 pp. 559- (1990)
- Saidu M. Waziri and Esam Z. Hamad "Volume-Explicit Equation of State for Fused Hard Sphere Chain Fluids", Industrial & Engineering Chemistry Research 47 pp. 9658-9662 (2008)
See also
References
- M. Whittle and A. J. Masters "Liquid crystal formation in a system of fused hard spheres", Molecular Physics 72 pp. 247-265 (1991)
- Carl McBride, Carlos Vega, and Luis G. MacDowell "Isotropic-nematic phase transition: Influence of intramolecular flexibility using a fused hard sphere model" Physical Review E 64 011703 (2001)
- Carl McBride and Carlos Vega "A Monte Carlo study of the influence of molecular flexibility on the phase diagram of a fused hard sphere model", Journal of Chemical Physics 117 pp. 10370-10379 (2002)
- Yaoqi Zhou, Carol K. Hall and George Stell "Thermodynamic perturbation theory for fused hard-sphere and hard-disk chain fluids", Journal of Chemical Physics 103 pp. 2688-2695 (1995)
- T. Boublík, C. Vega, and M. Diaz-Peña "Equation of state of chain molecules", Journal of Chemical Physics 93 pp. pp. 730-736 (1990)
- Antoine Chamoux and Aurelien Perera "On the linear hard sphere chain fluids", Molecular Physics '93 pp. 649-661 (1998)

![{\displaystyle m_{\rm {effective}}={\frac {[1+(m-1)L^{*}]^{3}}{[1+(m-1)L^{*}(3-L^{*2})/2]^{2}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/87c971dcd843e41062d341bfbcd501f4edea8001)




![{\displaystyle Z_{\mathrm {FHSC} }=1+(1+3\alpha )\eta _{0}(P^{*})+C_{\rm {FHSC}}[\eta _{0}(P^{*})]^{1.83}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/91afdc922fdfa04c21cdd02b43bebbdcfee850a4)
![{\displaystyle C_{\rm {FHSC}}=5.66\alpha (1-0.045[\alpha -1]^{1/2}\eta _{0})}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4149ca3dad157caa5ae90ae3f5937fe4ef96457e)

![{\displaystyle Z_{\mathrm {FHSC} }=1+4m_{\mathrm {eff} }P^{*}+{\frac {3}{4}}m_{\mathrm {eff} }P^{*}\ln \left[{\frac {3+P^{*}}{3+25P^{*}}}\right]+{\frac {216(m_{\mathrm {eff} }-1)P^{*}}{(3+P^{*})(3+25P^{*})\{16+3\ln[(3+P^{*})/(3+25P^{*})]\}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/14883bcaf0a2995db20fbd4f59e80d64d62d9a07)
